This is our Friday rubric: every week a new Science Page from the Bob Morrison’s Swine Health Monitoring Project. The previous editions of the science page are available on our website.
In today’s Science Page researchers at the USDA share the findings of a project assessing the virulence of an African Swine Fever virus strain isolated in the Dominican Republic in 2021. Thank you to reseachers Ramirez-Medina, O’Donnell, Silva, Espinoza, Velazquez-Salinas, Moran, Daite, Barrette, Faburay, Holland, Gladue, and Borca who worked on these experiments.
Main Points:
- The ASFV-DR21 strain clinical diseases varies according to route of exposure.
- Degree of clinical signs coincides with viral titer detection level in the collected samples.
- Detection of infectious virus and viral genome were inconsistent in nasal and oral swabs.
Introduction: This project was performed to describe the clinical presentation of domestic pigs inoculated with the ASFV strain isolated from a field sample in the province of Espaillat in the DR outbreak in July 2021. This particular isolate belongs to genotype II and is 99.99% identical to the Georgia 2007 strain.
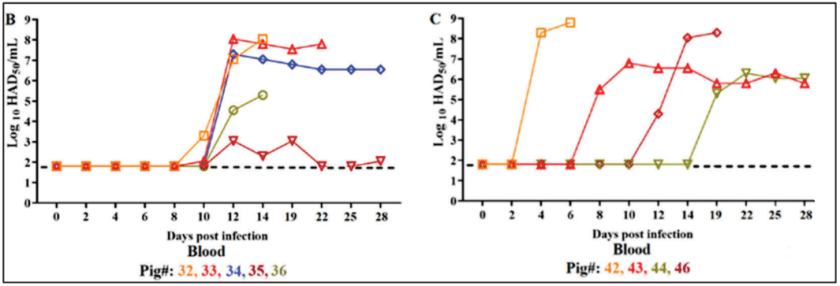
Materials and Methods: Three experiments were conducted, and in all experiments, 80-90 pound commercial Yorkshire crossbred female swine were used. For the first experiment, 5 pigs were inoculated intramuscularly (IM) ASFV-DR21 strain. In the second experiment, 5 pigs were inoculated oronasally (ON) with ASFV-DR21 strain. In the third experiment 5 non-infected pigs were co-housed with the IM-inoculated pigs, 24 hours post-inoculation. Blood, nasal and oral swabs were collected on day 2, 4, 6, 8, 10, 12, 14, 19, 22, 25, and 28 pi for detection for the presence of virus by qPCR, virus titrations, and ASFV-specific antibodies via ELISA. All animals that were euthanized, were done at humane clinical endpoints. The pigs that were not euthanized due to clinical endpoints were euthanized at the end of the study on day 28.
Results: Clinical signs for pigs with IM inoculation included fever (>104°F) by day 5 pi, along with depression, anorexia, and red-purple coloration of the skin. Clinical signs for pigs with ON inoculation and IM-contact were more varied. Some showed acute and fatal disease progression quickly, while others did not until day 12 and 13 pi or had a form that was not as acute. Finally, there were 3 pigs that didn’t develop any clinical signs, other than a fever. Viremia titers aligned with clinical presentation for those who developed clinical disease. Those who survived infection had variable titers (Figure 1). For IM inoculated pigs, low to moderate virus titers were detected in nasal swabs. For ON inoculated pigs, only 1 pig had a very low level of virus titer in a nasal swab. For IM-contact pigs, low-level viral titers in nasal swabs were found for 2 pigs on 2 different days. All oral swabs for all pigs, had no detectable viral titers. Ct values for detectable viral genome for nasal swabs are shown in Figure 2.
Discussion and Conclusion: The results highlight the inconsistent production of acute and fatal clinical disease progression of pigs that are not IM inoculated with ASFV-DR21 in a controlled laboratory setting. These observations are in agreement with the description of field cases of ASF in the DR, and a sharp contrast to the demonstrated virulence of ASFV Georgia 2010 isolate. IM inoculation of ASFV-DR21 does produce a rapid clinical disease, indicating that the virus is able to disseminate systemically. Animals that were in direct contact with IM inoculated animals showed a delayed development of symptoms. With viral shedding detected via nasal swabs in IM inoculated pigs at day 6 pi and all IM inoculated animals euthanized by day 7 dpi, it is estimated that contact pigs were infected approximately 5 to 6 days pi and took 5 to 7 days for viral infection to occur. No detection of infectious virus in oral swabs for all animals was surprising given previous observations of oral-nasal secretions of ASFV in infected animals. It should be noted that three pigs that developed high level viremia did not develop fatal disease, this could be attributed to the presence of antibodies. The high level of antibodies is comparable to the amount produced by live-attenuated experimental vaccines. Antibody production by surviving animals of ASFV-DR21 is thereby an important factor in control and surveillance strategies for the ASFV outbreak in the DR.

The full report can be found at: https://doi.org/10.3390/v14051090
